Why nobody is happy with FDA ruling on Plan B
Loading...
| Washington
The Food and Drug Administration’s decision to lower the age of eligibility for the morning-after pill – but not eliminate restrictions altogether – has left nobody completely satisfied.
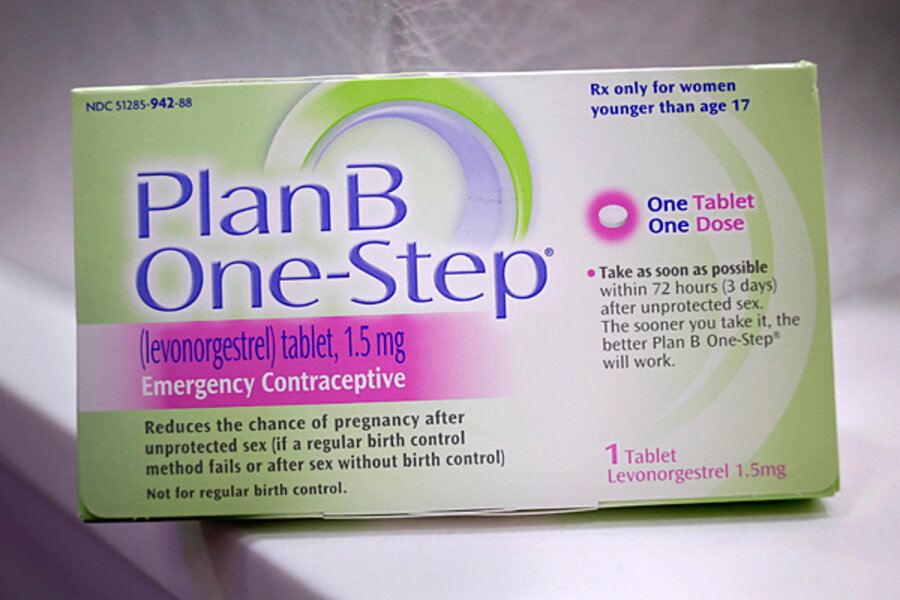
On Tuesday, the FDA announced that the emergency contraceptive known as Plan B One-Step would now be available, without prescription, to those ages 15 and older. That represents a shift from the current regulation, which made the pill available without a prescription only to people 17 and above. The new rule also allows the pill to be sold on pharmacy shelves, instead of being locked behind a pharmacy counter.
The FDA’s move came in response to an amended application by the pill’s manufacturer, Teva Women’s Health, which was filed after a 2011 decision by the FDA to restrict access to Plan B One-Step to women 17 and older. Tuesday's announcement was not a response to a federal judge’s ruling on April 4, which ordered the pill be made available within 30 days for all ages and without a prescription. That deadline is next Monday.
The Department of Justice is considering next steps on the litigation, according to the FDA. If the Justice Department opts not to appeal the federal judge’s order, it remains unclear what would happen with the conflicting new FDA rule, which goes into effect immediately.
The issue has long been politically charged. In 2011, Health and Human Services Secretary Kathleen Sebelius set an age restriction of 17 years on access to the morning-after pill, despite an FDA determination that the drug was safe and effective for all ages.
Reaction to the new FDA announcement from different actors in the longstanding debate over reproductive rights ranged from tepid support to outright hostility.
Cecile Richards, president of the Planned Parenthood Federation of America, called the new FDA rule “an important step forward to expand access to emergency contraception and for preventing unintended pregnancy.”
But, she added, “we continue to believe that the administration should lift all unnecessary restrictions to emergency contraception, consistent with the prevailing science and medicine.”
On the conservative side, Penny Nance, chief executive officer and president of Concerned Women for America, slammed the rule.
"It makes no sense that kids need parental permission to take aspirin at school, but they're free to buy and administer Plan B,” Ms. Nance said in a statement. “Also known as the morning-after pill, Plan B contains 40 times the dosage of Levonorgestrel, the same drug used in other forms of birth control. If Plan B is so 'safe,' then why is a prescription required for birth control?”
In the FDA announcement on the new rule, FDA Commissioner Margaret Hamburg, MD, cited research that indicates women 15 and older were able to understand how Plan B One-Step works, how to use it properly, and that it does not prevent the spread of sexually transmitted diseases.
Plan B One-Step is a single-dose pill that is most effective in decreasing the possibility of unwanted pregnancy if taken within three days of unprotected sexual intercourse, the FDA says.
The agency requires that the product carry a label that now reads, “not for sale to those under 15 years of age *proof of age required* not for sale where age cannot be verified.”
As opponents of the new rule point out, the age restriction could be easy to circumvent. Anyone – man or woman – who meets the age requirement can now purchase the drug over the counter for an underage woman.
When asked what constitutes proof of age for a 15-year-old, an FDA spokeswoman replied, “passport or birth certificate.”
Some groups that support widespread availability of Plan B also expressed hostility to the new FDA rule. The Center for Reproductive Rights (CRR) – which won a victory last month when federal Judge Edward Korman ordered that Plan B be made available without age or point-of-sale restrictions – asserted that any restrictions will keep the drug out of the hands of women who need it.
“Lowering the age restriction to 15 for over-the-counter access to Plan B One-Step may reduce delays for some young women – but it does nothing to address the significant barriers that far too many women of all ages will still find if they arrive at the drugstore without identification or after the pharmacy gates have been closed for the night or weekend,” the CRR said.
The group pledged to continue its battle in court.